Photo from wikipedia
To the Editor: Pruritus is a severely distressing symptom that accompanies cholestatic liver diseases in children. In this study, we evaluated a new emollient mixture for pruritus in cholestatic children.… Click to show full abstract
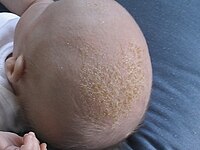
To the Editor: Pruritus is a severely distressing symptom that accompanies cholestatic liver diseases in children. In this study, we evaluated a new emollient mixture for pruritus in cholestatic children. We recruited 51 children (1–10 y), who were compliant to systemic antipruritic drugs. Patients with end-stage-liver-disease, personal or family history of atopicdermatitis, or pruritus due to other systemic diseases, were excluded. All children were subjected to pruritus severity baseline evaluation, using 5-D-Itch-Scale (5-D) [1], and 12item-pruritus-severity-scale (12-PSS) [2]. We supplemented patients with a specifically-prepared topical mixture using coconut-oil with vitamins A and E (Final concentration 200,000 IU vitamin-A and 1600 IU vitamin-E/100 mL extra-virgin coconut-oil). Systemic therapy was not altered 2 mo before starting topical therapy and was maintained along. Caregivers were instructed to apply the emollient daily before sleep, after warm bath and over the whole body. Coconut-oil is a soothing agent with broad-spectrum antimicrobial activity [3]. Vitamin-A is necessary to maintain skin integrity and sebum production, while vitamin-E confers photo-protective activity against erythema, edema, and sunburn cell-formation [4]. Initial dermatological assessment revealed xerosis, excoriations, lichenification, prurigo and scalp baldness in 51,46,22,18 and 4 patients, respectively. Cholestatic children with normal GGT (n = 20) had higher scores of both 5-D: 15.7 ± 7.1 (median 15; quartiles 9.5–22.5) and 12-PSS: 13.7 ± 5.2 (13; 10.5–18) points compared to those with high GGT (n = 31) 12.8 ± 5.3 (12; 8.5–17.5) and 11.5 ± 4.3 (11; 9–15) points (p = 0.161 and 0.080, respectively). After 3 mo, the topical therapy improved xerosis in 100% of evaluated children (33 evaluated on follow-up), and scalp baldness in 2/4 patients (50%). It also reduced absolute values for 5-D and 12PSS scores in the follow-up cohort from 14.2 ± 5.6 (14; 9–20) and 12.7 ± 4.1 (13; 10–16) points initially to 12.5 ± 5.8 (13; 8– 15) and 10.9 ± 4.4 (11; 8–13) points after therapy (p = 0.226 and 0.054, respectively). Using the 5-D score, pruritus was deemed severe (≥18 points) in 10 patients (30.3%) initially. This number decreased to six (18.1%) after 3 mo of topical treatment (p = 0.210). Similarly, the 12-PSS score showed a decline in severely affected patients (≥12 points) from 20 patients (60.6%) to 11 (33.3%) (p = 0.026). Coconut-oil was previously used in atopic dermatitis [5]; however, to our knowledge it was not reported in systemic disease pruritus. Coconut-oil with vitamins A/E could be a supplementary treatment for pruritus in cholestatic children as it may help systemic drugs to achieve optimal response before shifting to other therapeutic modalities.
Journal Title: Indian Journal of Pediatrics
Year Published: 2021
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!