Photo from wikipedia
While the original protein Toll in Drosophila melanogaster regulates both host defense and morphogenesis, the role of its ortholog Toll-like receptors (TLRs), the interleukin 1 receptor (IL-1R) family, and the… Click to show full abstract
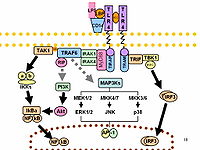
While the original protein Toll in Drosophila melanogaster regulates both host defense and morphogenesis, the role of its ortholog Toll-like receptors (TLRs), the interleukin 1 receptor (IL-1R) family, and the associated signaling pathways in mammalian brain development and structure is poorly understood. Because the adaptor protein myeloid differentiation primary response protein 88 (MyD88) is essential for downstream signaling of most TLRs and IL-1R, we systematically investigated the effect of MyD88 deficiency on murine brain structure during development and on behavior. In neonatal Myd88-/- mice, neocortical thickness was reduced, while density of cortical neurons was increased. In contrast, microglia, astrocyte, oligodendrocyte, and proliferating cell numbers were unchanged in these mice compared to wild-type mice. In adult Myd88-/- mice, neocortical thickness was unaltered, but neuronal density in neocortex and hippocampus was increased. Neuron arborization was less pronounced in adult Myd88-/- mice compared to wild-type animals. In addition, numbers of microglia and proliferating cells were increased in the neocortex and subventricular zone, respectively, with unaltered astrocyte and oligodendrocyte numbers, and myelinization was enhanced in the adult Myd88-/- neocortex. These morphologic changes in the brain of adult Myd88-/- mice were accompanied by specific behavioral traits, such as decreased locomotor activity, increased anxiety-like behavior, but normal day/light activity, satisfactory learning, short- and long-term spatial memory, potential cognitive inflexibility, and increased hanging and locomotor behavior within their home cage. Taken together, MyD88 deficiency results in morphologic and cellular changes in the mouse brain, as well as in altered natural and specific behaviors. Our data indicate a pathophysiological significance of MyD88 for mammalian CNS development, structure, and function.
Journal Title: Brain, Behavior, and Immunity
Year Published: 2021
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!