Photo from wikipedia
Bone morphogenetic proteins (BMPs) are known to induce new bone formation in vivo but treating trabecular bone defects with a BMP based therapeutic remains controversial. Here, we evaluated the safety… Click to show full abstract
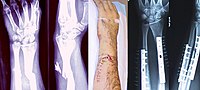
Bone morphogenetic proteins (BMPs) are known to induce new bone formation in vivo but treating trabecular bone defects with a BMP based therapeutic remains controversial. Here, we evaluated the safety and efficacy of a novel Autologous Bone Graft Substitute (ABGS) comprised of recombinant human BMP6 (rhBMP6) dispersed within an autologous blood coagulum (ABC) as a physiological natural carrier in patients with a closed distal radial fracture (DRF). We enrolled 32 patients in a randomized, standard of care (SoC) and placebo (PBO) controlled, double-blinded Phase I First in Human (FiH) clinical trial. ABGS was prepared from peripheral blood as 250 μg rhBMP6/mL ABC or PBO (1 mL ABC containing excipients only) and was administered dorsally via a syringe injection into the fracture site following closed fracture fixation with 3 Kirschner wires. Patients carried an immobilization for 5 weeks and were followed-up for 0 to 26 weeks by clinical examination, safety, serial radiographic analyses and CT. During the 13 weeks follow-up and at 26 weeks post study there were no serious adverse reactions recorded. The results showed that there were no detectable anti-rhBMP6 antibodies in the blood of any of the 32 patients at 13- and 26-weeks following treatment. Pharmacokinetic analyses of plasma from patients treated with ABGS showed no detectable rhBMP6 at any time point within the first 24 hours following administration. The CT image and radiographic analyses score from patients treated with AGBS showed significantly accelerated bone healing as compared to PBO and SoC at 5 and 9 weeks (with high effect sizes and P=0.027), while at week 13 all patients had similar healing outcomes. In conclusion, we show that intraosseous administration of ABGS (250 μg rhBMP6/mL ABC) into the distal radial fracture site demonstrated a good tolerability with no serious adverse reactions as well as early accelerated trabecular bone healing as compared to control PBO and SoC patients.
Journal Title: Bone
Year Published: 2020
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!