Photo from wikipedia
Abstract The influence of methanol solvent in assisting the nucleation of adipic acid crystals has been investigated for the first time. The pH value and nucleation period was determined. The… Click to show full abstract
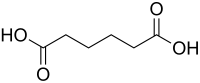
Abstract The influence of methanol solvent in assisting the nucleation of adipic acid crystals has been investigated for the first time. The pH value and nucleation period was determined. The better- quality adipic acid single crystals having crystal size up to 10 × 7 x 6mm3 were grown successfully by conventional slow solvent evaporation experiment at room temperature and their solubility has been experimented with four different solvents namely methanol, ethanol, acetone and water at three different temperature 30°, 40° and 50° respectively. Crystallographic information, crystalline nature and characteristic h k l planes of adipic acid was revealed by single crystal XRD and powder XRD studies. The direct optical band gap energy, characteristic functional groups and other information like thermal decomposition temperature, melting point were studied from UV –Vis-NIR, Spectroscopic FTIR and TG-DTA instrument analysis. The measured powder SHG efficiency of adipic acid is found to be 2.14 times superior to the reference material potassium dihydrogen phosphate (KDP).
Journal Title: Optik
Year Published: 2021
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!