Photo from wikipedia
Abstract In this work, we measured the vapor-liquid equilibrium of 2,3,3,3-tetrafluoroprop-1-ene (R1234yf) in three ionic liquids, including 1-butyl-3-methylimidazolium hexafluorophosphate ([Bmim][PF6]), 1-hexyl-3-methylimidazolium hexafluorophosphate ([Hmim][PF6]), and 1-octyl-3-methylimidazolium hexafluorophosphate ([Omim][PF6]). The phase equilibrium… Click to show full abstract
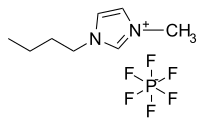
Abstract In this work, we measured the vapor-liquid equilibrium of 2,3,3,3-tetrafluoroprop-1-ene (R1234yf) in three ionic liquids, including 1-butyl-3-methylimidazolium hexafluorophosphate ([Bmim][PF6]), 1-hexyl-3-methylimidazolium hexafluorophosphate ([Hmim][PF6]), and 1-octyl-3-methylimidazolium hexafluorophosphate ([Omim][PF6]). The phase equilibrium data were reported from 283.15 K to 343.15 K, and the measurements were carried out based on the isochoric method. The experimental data were correlated by the nonrandom two-liquid (NRTL) activity coefficient model. The absolute average relative deviations of pressure between experimental data and calculated values are 2.06%, 1.66%, and 1.55% for R1234yf/[Bmim][PF6], R1234yf/[Hmim][PF6], and R1234yf/[Omim][PF6] systems, respectively. The maximum relative deviations are 5.59%, 4.87%, and 4.72%.
Journal Title: Journal of Molecular Liquids
Year Published: 2018
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!