Photo from wikipedia
Abstract Imipramine hydrochloride ([H-IMI]Cl), C19H24N2.HCl, is the prototypic tricyclic antidepressant (TCA) inhibitor of norepinephrine and serotonin neuronal reuptake. The molecular structure, molecular electrostatic potential (MEP), natural bond orbital (NBO) analysis,… Click to show full abstract
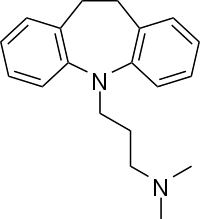
Abstract Imipramine hydrochloride ([H-IMI]Cl), C19H24N2.HCl, is the prototypic tricyclic antidepressant (TCA) inhibitor of norepinephrine and serotonin neuronal reuptake. The molecular structure, molecular electrostatic potential (MEP), natural bond orbital (NBO) analysis, linear and non-linear optical (NLO) properties of [H-IMI]Cl have been investigated using the density functional theory (DFT) calculations with the B3LYP level at the 6‒311++G(d,p) basis set. The UV–Vis spectra for [H-IMI]Cl were experimentally studied in water and methanol. TD‒DFT calculations in water and methanol were employed to investigate the absorption wavelengths (λ), excitation energies (E), and oscillator strengths (f) for the UV–Vis analysis and the major contributions to the electronic transitions. From NBO analysis, the orbitals with the stabilization energy E(2) of 192.15 kcal/mol are π*(C5 C18) as donor NBO and π*(C19 C20) as acceptor NBO. The FT‒IR (4000‒400 cm−1) and FT‒Raman (3500–50 cm−1) spectra have been measured and analyzed. The assignment of bands observed vibrational spectra have been made by comparison of its calculated theoretical vibrational frequencies obtained using the DFT/B3LYP/6‒311++G(d,p) method. The detailed vibrational assignments were performed with the DFT calculation, and the potential energy distribution (PED) of [H-IMI]Cl was obtained by the Vibrational Energy Distribution Analysis 4 (VEDA4) program. The scaled frequencies resulted in good agreement with the observed spectral patterns.
Journal Title: Journal of Molecular Structure
Year Published: 2018
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!