Photo from wikipedia
ConspectusAlkyl and alkenyl arenes are of substantial value in both large-scale and fine chemical processes. Billions of pounds of alkyl and alkenyl arenes are produced annually. Historically, the dominant method… Click to show full abstract
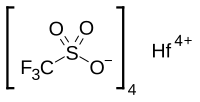
ConspectusAlkyl and alkenyl arenes are of substantial value in both large-scale and fine chemical processes. Billions of pounds of alkyl and alkenyl arenes are produced annually. Historically, the dominant method for synthesis of alkyl arenes is acid-catalyzed arene alkylation, and alkenyl arenes are often synthesized in a subsequent dehydrogenation step. But these methods have limitations that result from the catalytic mechanism including (1) common polyalkylation, which requires an energy intensive transalkylation process, (2) quantitative selectivity for Markovnikov products for arene alkylation using α-olefins, (3) for substituted arenes, regioselectivity that is dictated by the electronic character of the arene substituents, (4) inability to form alkenyl arenes in a single process, and (5) commonly observed slow reactivity with electron-deficient arenes. Transition-metal-catalyzed aryl-carbon coupling reactions can produce alkyl or alkenyl arenes from aryl halides. However, these reactions often generate halogenated waste and typically require a stoichiometric amount of metal-containing transmetalation reagent. Transition-metal-catalyzed arene alkylation or alkenylation that involves arene C-H activation and olefin insertion into metal-aryl bonds provides a potential alternative method to prepare alkyl or alkenylation arenes. Such reactions can circumvent carbocationic intermediates and, as a result, can overcome some of the limitations mentioned above. In particular, controlling the regioselectivity of the insertion of α-olefins into metal-aryl bonds provides a strategy to selectively synthesize anti-Markovnikov products. But, previously reported catalysts often show limited longevity and low selectivity for anti-Markovnikov products.In this Account, we present recent developments in single-step arene alkenylation using Rh catalyst precursors. The reactions are successful for unactivated hydrocarbons and exhibit unique selectivity. The catalytic production of alkenyl arenes operates via Rh-mediated aromatic C-H activation, which likely occurs by a concerted metalation-deprotonation mechanism, olefin insertion into a Rh-aryl bond, β-hydride elimination from the resulting Rh-hydrocarbon product, and net dissociation of alkenyl arene with formation of a Rh hydride. Reaction of the Rh hydride with Cu(II) oxidant completes the catalytic cycle. Although Rh nanoparticles can be formed under some conditions, mechanistic studies have revealed that soluble Rh species are likely responsible for the catalysis. These Rh catalyst precursors achieve high turnovers with >10,000 catalytic turnovers observed in some cases. Under anaerobic conditions, Cu(II) carboxylates are used as the oxidant. In some cases, aerobic recycling of Cu(II) oxidant has been demonstrated. Hence, the Rh arene alkenylation catalysis bears some similarities to Pd-catalyzed olefin oxidation (i.e., the Wacker-Hoechst process). The Rh-catalyzed arene alkenylation is compatible with some electron-deficient arenes, and they are selective for anti-Markovnikov products when using substituted olefins. Finally, when using monosubstituted arenes, consistent with a metal-mediated C-H activation process, Rh-catalyzed alkenylation of substituted arenes shows selectivity for meta- and para-alkenylation products.
Journal Title: Accounts of chemical research
Year Published: 2020
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!