Photo from wikipedia
Phenylalanine ammonia lyase (PAL) catalyzes the reversible conversion of l-phenylalanine into the corresponding trans-cinnamic acid, providing a route to optically pure α-amino acids. We explored the catalytic function of all… Click to show full abstract
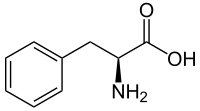
Phenylalanine ammonia lyase (PAL) catalyzes the reversible conversion of l-phenylalanine into the corresponding trans-cinnamic acid, providing a route to optically pure α-amino acids. We explored the catalytic function of all five PALs encoded in the genome of lettuce (Lactuca sativa L.) that are previously known to be involved in wound browning. All LsPALs were active toward l-phenylalanine in the ammonia elimination reaction and displayed maximum activity at 55-60 °C and pH 9.0-9.5. However, four of them, LsPAL1-LsPAL4, showed significantly higher activity and thermal stability than LsPAL5, as well as a broader substrate spectrum including some challenging substrates with steric demanding or electron-donating substituents. The best one LsPAL3 was subjected to the kinetic resolution of a panel of 21 rac-phenylalanine derivatives, as well as the ammonia addition of 21 cinnamic acid derivatives. It showed excellent enantioselectivity in most cases and significantly better activity than previously described PALs for a number of challenging non-natural substrates, demonstrating its great potential in biocatalysis.
Journal Title: Journal of agricultural and food chemistry
Year Published: 2023
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!