Photo from wikipedia
We report the characterization of a series of novel phosphinidene-stabilized (P-stabilized) boryl cation radicals, in which the phosphinidene and boryl are stabilised by iPrNHC (iPrNHC[:C{N(iPr)C(H)}2]), and the P-stabilized boryl (P… Click to show full abstract
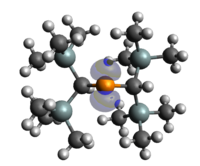
We report the characterization of a series of novel phosphinidene-stabilized (P-stabilized) boryl cation radicals, in which the phosphinidene and boryl are stabilised by iPrNHC (iPrNHC[:C{N(iPr)C(H)}2]), and the P-stabilized boryl (P → B) moieties are linked by 1,8-naphthalene (1PB-a), 1,10-biphenyl (1PB-b), 1,2-perylene (1PB-c), and 4,5-perylene (1PB-d), to form a series of 1PB compounds. The 2PB series is designed by adding another P-stabilized boryl (P → B) unit into the 1PB series, in which the two P-stabilized boryl (P → B) moieties for each 2PB compound are linked by 1,4,5,8-naphthalene, (2PB-a), 1,5,6,10-biphenyl (2PB-b), 1,2,7,8-perylene (2PB-c), and 4,5,10,11-perylene (2PB-d), respectively. Theoretical calculations demonstrate that for all the studied molecules, the spin density mainly locates on the B atoms. Interestingly, the series of 2PB(a-d) compounds possess SOMO-HOMO conversion properties, while 1PB(a-d) compounds obey the Aufbau principle, resulting from the difference in the number of the P-stabilized boryl (P → B) moieties and an increase of the π-conjugation bridge that lead to the significantly increased HOMO energy in 2PB(a-d) compounds, which should be responsible for the different structural properties of compounds 1PB(a-d) and 2PB(a-d). The natural bond orbital (NBO) and atoms in molecules (AIM) analysis reveal how the interactions contribute to the covalent bond between P and B atoms. Moreover, the absorption properties show that the spectra of the 2PB(a-d) compounds are red-shifted relative to those of the corresponding 1PB(a-d) compounds in the near infrared region. We hope this work can provide new insights into tuning the electronic structures of the well-defined forms of P-stabilized boryl cation radicals and expand their potential application in organic optoelectronics.
Journal Title: Dalton transactions
Year Published: 2023
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!