Photo from wikipedia
The thiazolidinedione class PPARγ agonists as antidiabetic agents are restricted in clinical use because of the side effects such as edema, weight gain, and heart failure. The single and selective… Click to show full abstract
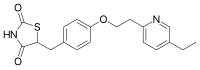
The thiazolidinedione class PPARγ agonists as antidiabetic agents are restricted in clinical use because of the side effects such as edema, weight gain, and heart failure. The single and selective agonism of PPARγ is the main cause of side effects. The multi-target cooperative PPARα/γ dual agonist development is a hot topic in the antidiabetic medicinal chemistry field. Saroglitazar is the first approved PPARα/γ dual agonist, available in India for the treatment of diabetic dyslipidemia. It got rid of these side effects. With the aim of finding more protent PPARα/γ dual agonists, the scaffold hopping was used to replace α-o phenylpropionic acid skeleton of saroglitazar with L-tyrosine skeleton. Then, the structural modification was carried out designing 72 compounds. Considering the importance of chirality, opposite configuration of 72 compounds was also studied. 12 compounds with better -cdocker energy were screened by molecular docking. Subsequently, the pharmacokinetic properties and toxicity evaluated by ADMET prediction, 11 of them showed better properties. Comp#L-17-1 and comp#L-3-1 were regarded as representatives to study the binding stability by molecular dynamics (MD) simulations. The MD simulation results of comp#L-17-1-PPARs (α, γ) and comp#L-3-1-PPARs (α, γ) provided structure reference for the research and development of novel PPARα/γ dual agonists.
Journal Title: Journal of Biomolecular Structure and Dynamics
Year Published: 2018
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!