Photo from wikipedia
Abstract The multifunctional human DNA2 (hDNA2) nuclease/helicase is required to process DNA ends for homology-directed recombination repair (HDR) and to counteract replication stress. To participate in these processes, hDNA2 must… Click to show full abstract
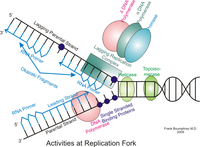
Abstract The multifunctional human DNA2 (hDNA2) nuclease/helicase is required to process DNA ends for homology-directed recombination repair (HDR) and to counteract replication stress. To participate in these processes, hDNA2 must localize to the nucleus and be recruited to the replication or repair sites. However, because hDNA2 lacks the nuclear localization signal that is found in its yeast homolog, it is unclear how its migration into the nucleus is regulated during replication or in response to DNA damage. Here, we report that the E3 ligase TRAF6 binds to and mediates the K63-linked polyubiquitination of hDNA2, increasing the stability of hDNA2 and promoting its nuclear localization. Inhibiting TRAF6-mediated polyubiquitination abolishes the nuclear localization of hDNA2, consequently impairing DNA end resection and HDR. Thus, the current study reveals a mechanism for the regulation of hDNA2 localization and establishes that TRAF6-mediated hDNA2 ubiquitination activates DNA repair pathways to maintain nuclear genome integrity.
Journal Title: Nucleic Acids Research
Year Published: 2019
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!