Photo from wikipedia
In kidney transplant patients, clinically relevant drug–drug interactions (DDIs) with immunosuppressants potentially lead to serious adverse drug events (ADEs). The aim of this study was (i) to show that five… Click to show full abstract
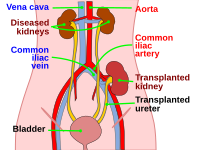
In kidney transplant patients, clinically relevant drug–drug interactions (DDIs) with immunosuppressants potentially lead to serious adverse drug events (ADEs). The aim of this study was (i) to show that five clinical decision support systems (CDSSs) differ in their ability to identify clinically relevant potential DDIs (pDDIs) of immunosuppressants in kidney transplant patients and (ii) to compare CDSSs in terms of their ability to identify clinically relevant pDDIs in this context.
Journal Title: Journal of Clinical Pharmacy and Therapeutics
Year Published: 2017
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!