Photo from wikipedia
Introduction Among critically ill patients undergoing orotracheal intubation in the emergency department (ED) or intensive care unit (ICU), failure to visualise the vocal cords and intubate the trachea on the… Click to show full abstract
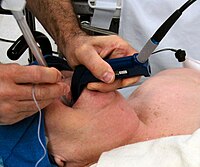
Introduction Among critically ill patients undergoing orotracheal intubation in the emergency department (ED) or intensive care unit (ICU), failure to visualise the vocal cords and intubate the trachea on the first attempt is associated with an increased risk of complications. Two types of laryngoscopes are commonly available: direct laryngoscopes and video laryngoscopes. For critically ill adults undergoing emergency tracheal intubation, it remains uncertain whether the use of a video laryngoscope increases the incidence of successful intubation on the first attempt compared with the use of a direct laryngoscope. Methods and analysis The DirEct versus VIdeo LaryngosCopE (DEVICE) trial is a prospective, multicentre, non-blinded, randomised trial being conducted in 7 EDs and 10 ICUs in the USA. The trial plans to enrol up to 2000 critically ill adults undergoing orotracheal intubation with a laryngoscope. Eligible patients are randomised 1:1 to the use of a video laryngoscope or a direct laryngoscope for the first intubation attempt. The primary outcome is successful intubation on the first attempt. The secondary outcome is the incidence of severe complications between induction and 2 min after intubation, defined as the occurrence of one or more of the following: severe hypoxaemia (lowest oxygen saturation <80%); severe hypotension (systolic blood pressure <65 mm Hg or new or increased vasopressor administration); cardiac arrest or death. Enrolment began on 19 March 2022 and is expected to be completed in 2023. Ethics and dissemination The trial protocol was approved with waiver of informed consent by the single institutional review board at Vanderbilt University Medical Center and the Human Research Protection Office of the Department of Defense. The results will be presented at scientific conferences and submitted for publication in a peer-reviewed journal. Trial registration number ClinicalTrials.gov Registry (NCT05239195).
Journal Title: BMJ Open
Year Published: 2022
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!