Photo from wikipedia
Primary aldosteronism affects ≈5% to 10% of hypertensive patients and has unilateral and bilateral forms. Most unilateral primary aldosteronism is caused by computed tomography-detectable aldosterone-producing adenomas, which express CYP11B2 (aldosterone… Click to show full abstract
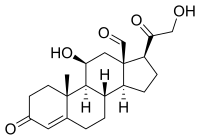
Primary aldosteronism affects ≈5% to 10% of hypertensive patients and has unilateral and bilateral forms. Most unilateral primary aldosteronism is caused by computed tomography-detectable aldosterone-producing adenomas, which express CYP11B2 (aldosterone synthase) and frequently harbor somatic mutations in aldosterone-regulating genes. The cause of the most common bilateral form of primary aldosteronism, idiopathic hyperaldosteronism (IHA), is believed to be diffuse hyperplasia of aldosterone-producing cells within the adrenal cortex. Herein, a multi-institution cohort of 15 IHA adrenals was examined with CYP11B2 immunohistochemistry and next-generation sequencing. CYP11B2 immunoreactivity in adrenal glomerulosa harboring non-nodular hyperplasia was only observed in 4/15 IHA adrenals suggesting that hyperplasia of CYP11B2-expressing cells may not be the major cause of IHA. However, the adrenal cortex of all IHA adrenals harbored at least 1 CYP11B2-positive aldosterone-producing cell cluster (APCC) or micro-aldosterone-producing adenomas. The number of APCCs per case (and individual APCC area) in IHA adrenals was significantly larger than in normotensive controls. Next-generation sequencing of DNA from 99 IHA APCCs demonstrated somatic mutations in genes encoding the L-type calcium voltage-gated channel subunit &agr; 1-D (CACNA1D, n=57; 58%) and potassium voltage-gated channel subfamily J-5 (KCNJ5, n=1; 1%). These data suggest that IHA may result from not only hyperplasia but also the accumulation or enlargement of computed tomography-undetectable APCC harboring somatic aldosterone-driver gene mutations. The high prevalence of mutations in the CACNA1D L-type calcium channel provides a potential actionable therapeutic target that could complement mineralocorticoid blockade and inhibit aldosterone overproduction in some IHA patients.
Journal Title: Hypertension
Year Published: 2018
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!