Photo from wikipedia
Antibiotic resistance is one of the greatest contemporary threats to the human health, which has increasingly been undermining the effectiveness of existing antimicrobial therapies. Development of novel antibiotics is inarguably… Click to show full abstract
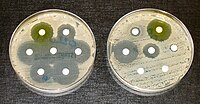
Antibiotic resistance is one of the greatest contemporary threats to the human health, which has increasingly been undermining the effectiveness of existing antimicrobial therapies. Development of novel antibiotics is inarguably the key to combat this threat, yet this is a lengthy process (10 to 15 years) at a hefty price tag of approximately US$1.3 billion for development of an approved drug [1,2]. A new drug, once introduced into the market, also faces the risk of drug resistance emergence. It is, therefore, essential to diversify therapeutic strategies, including revival and reintroduction of “old” antibacterials for treating multidrug-resistant pathogens [3]. Nitrofuran class of synthetic molecules, introduced in the 1940s and 1950s, belongs to this category [4]. Several nitrofurans are currently on the market: nitrofurazone for topical infections and urinary catheter coating, nitrofurantoin for urinary tract infections, and furazolidone for bacterial diarrhea and Helicobacter pylori infections. Here, we highlight aspects of this drug class that have recently been unraveled, laying foundation for future improvements and judicial uses of nitrofurans against ever-expanding antibiotic-resistant bacteria.
Journal Title: PLoS Pathogens
Year Published: 2021
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!