Photo from wikipedia
Abstract Room temperature ionic liquids (RTILs) represent a recent new class of solvents with potential application in liquid/liquid extraction based nuclear fuel reprocessing due to their unique physical and chemical… Click to show full abstract
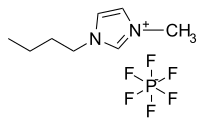
Abstract Room temperature ionic liquids (RTILs) represent a recent new class of solvents with potential application in liquid/liquid extraction based nuclear fuel reprocessing due to their unique physical and chemical properties. The work herein provides a comparison of U(VI) extraction by 8-hydroxyquinoline (HOX) in a commonly used RTIL, i.e. 1-butyl-3-methylimidazolium hexafluorophosphate ([C4mim][PF6]) and in conventional solvent, i.e. dichloromethane (CH2Cl2). The effect of HOX concentration, solution acidity and nitrate ions on the extraction were discussed in detail, and the speciation analyses of the extracted U(VI) were performed. One of the main emphasis of this work is the extraction mechanism of U(VI) extracted from aqueous phase into RTILs and conventional solvent. In CH2Cl2, the extraction occurs through a combination of ion change and neutral complexation, and the extracted complex is proposed as UO2(OX)2HOX. In [C4mim][PF6], although a cation-change mechanism as previously reported for RTILs-based system was involved, the extracted complex of UO2(OX)1.5(HOX)1.5(PF6)0.5 gave a clear indication that the usage of HOX as an acidic extractant markedly inhibited the solubility loss of [C4mim][PF6] during the extraction by leaching H+ to aqueous phase. Moreover, the extracted U(VI) in [C4mim][PF6] can be easily stripped by using 0.01 M nitric acid, which provides a simple way of the ionic liquid recycling.
Journal Title: Radiochimica Acta
Year Published: 2017
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!