Photo from wikipedia
To quantitatively convert biomass into value-added chemicals or biofuels is still a great challenge. Herein, we develop an efficient electrochemical method to convert the biomass-derived valeric acid into ethers (butyl… Click to show full abstract
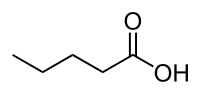
To quantitatively convert biomass into value-added chemicals or biofuels is still a great challenge. Herein, we develop an efficient electrochemical method to convert the biomass-derived valeric acid into ethers (butyl methyl ether and butyraldehyde dimethyl acetal) or esters (methyl valerate) on boron-doped diamond (BDD) electrode in methanol solution. The products distribution was well controlled by tuning the supporting electrolyte and the KOH amount. 98.7% selectivity and ca. 58% Faraday efficiency for ethers and 96% selectivity for esters were achieved by optimizing the supporting electrolytes, initial molar ratio of VA to KOH, VA feed concentration and anodic potential. Strong alkali as supporting electrolyte, high VA feed concentration and initial VA/KOH=1 (mol/mol) favoured the formation of ether products. Fatty acid methyl esters were high-selectively (>96%) produced by using NaClO4 as the supporting electrolyte and/or adding excessive KOH (initial KOH/VA ratio beyond 2:1). Moreover, partially replacing methanol with ethanol, only one new diether (1,1-diethoxybutane) was formed. A rational mechanism of synthetic butyraldehyde dimethyl acetal via electrocatalytic decarboxylation, rearrangement of carbenium ion, Aldol reaction on BDD in alcohols solution was developed. This strategy may provide a new direction for renewable chemicals and fuels from biomass.
Journal Title: International Journal of Electrochemical Science
Year Published: 2018
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!