Photo from wikipedia
In this study, sugarcane bagasse (SCB) was treated with sodium hydroxide and bleached to separate the non-cellulose components to obtain cellulose (CE) fibres. Cross-linked cellulose–poly(sodium acrylic acid) hydrogel (CE–PAANa) was… Click to show full abstract
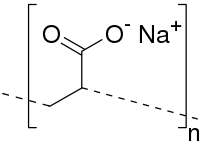
In this study, sugarcane bagasse (SCB) was treated with sodium hydroxide and bleached to separate the non-cellulose components to obtain cellulose (CE) fibres. Cross-linked cellulose–poly(sodium acrylic acid) hydrogel (CE–PAANa) was successfully synthesised via simple free-radical graft-polymerisation to remove heavy metal ions. The structure and morphology of the hydrogel display an open interconnected porous structure on the surface of the hydrogel. Various factors influencing batch adsorption capacity, including pH, contact time, and solution concentration, were investigated. The results showed that the adsorption kinetics were in good agreement with the pseudo-second-order kinetic model and that the adsorption isotherms followed the Langmuir model. The maximum adsorption capacities calculated by the Langmuir model are 106.3, 333.3, and 163.9 mg/g for Cu(II), Pb(II), and Cd(II), respectively. Furthermore, X-ray photoelectron spectroscopy (XPS) and energy-dispersive X-ray spectrometry (EDS) results demonstrated that cationic exchange and electrostatic interaction were the main heavy metal ions adsorption mechanisms. These results demonstrate that CE–PAANa graft copolymer sorbents from cellulose-rich SCB can potentially be used for the removal of heavy metal ions.
Journal Title: International Journal of Molecular Sciences
Year Published: 2023
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!