Photo from wikipedia
INTRODUCTION This study analyzes the safety and efficacy results of the Indian population subset from the RECLAIM trial investigating the non-inferiority of Ceftazidime-Avibactam (CAZ-AVI) plus metronidazole to meropenem and interprets… Click to show full abstract
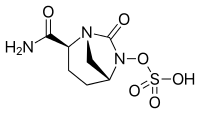
INTRODUCTION This study analyzes the safety and efficacy results of the Indian population subset from the RECLAIM trial investigating the non-inferiority of Ceftazidime-Avibactam (CAZ-AVI) plus metronidazole to meropenem and interprets its relevance. METHODOLOGY The study design, subjects inclusion criteria, dosage, safety and efficacy evaluations in Indian patients have been followed as per the RECLAIM trial protocol. RESULTS A total of 142 Indian patients with complicated intra-abdominal infection were enrolled across eight centers in India, 125 of them were randomized to either CAZ-AVI + metronidazole (n = 62) or meropenem (n = 63) group. the clinical cure rates in modified intention-to-treat (MITT; all randomized patients who met minimum disease requirements and received any amount of study drug) and clinically evaluable (CE , patients who had an evaluable assessment and no protocol deviations) analysis sets, was numerically comparable to the results of overall population for CAZ-AVI + metronidazole [MITT: 82.5% (Overall, n = 429/520) versus 89.3% (Indian, n = 50/56); CE: 91.7% (Overall, n = 376/410) versus 97.8% (Indian, n = 45/46)] and meropenem [MITT: 84.9% (Overall, n = 444/523) versus 84.7% (Indian, n = 50/59); CE: 92.5% (Overall, n = 385/416) versus 95.5% (Indian, n = 42/44)]. No new safety findings were reported in the Indian population. CONCLUSIONS CAZ-AVI + metronidazole proved to be an effective option for critical patients with complicated intra-abdominal infection and can be considered as an alternative to carbapenems in the ICU setting for the treatment of resistant pathogens.
Journal Title: Journal of infection in developing countries
Year Published: 2022
Link to full text (if available)
Share on Social Media: Sign Up to like & get
recommendations!