
Efficacy and safety of glecaprevir and pibrentasvir treatment for 8 or 12 weeks in patients with recurrent hepatitis C after liver transplantation: a Japanese multicenter experience
Sign Up to like & getrecommendations! Published in 2019 at "Journal of Gastroenterology"
DOI: 10.1007/s00535-019-01561-1
Abstract: BackgroundEfficacy of 8-week regimen with direct-acting antivirals (DAA) for patients with hepatitis C after liver transplantation has not been clarified. This study aimed to clarify the efficacy and safety of glecaprevir and pibrentasvir therapy for… read more here.
Keywords: hepatitis liver; treatment; liver; liver transplantation ... See more keywords

Effectiveness of 8-Week Glecaprevir/Pibrentasvir for Treatment-Naïve, Compensated Cirrhotic Patients with Chronic Hepatitis C Infection
Sign Up to like & getrecommendations! Published in 2020 at "Advances in Therapy"
DOI: 10.1007/s12325-020-01301-5
Abstract: Introduction Glecaprevir/pibrentasvir (G/P) was approved on 26 September 2019 by the US Food and Drug Administration for 8-week duration in treatment-naïve (TN) hepatitis C virus (HCV)-infected patients with compensated cirrhosis (CC). Evidence from the EXPEDITION-8… read more here.
Keywords: effectiveness week; week; treatment; treatment compensated ... See more keywords

SAFETY AND EFFICACY OF STATIN MANAGEMENT DURING GLECAPREVIR/PIBRENTASVIR TREATMENT FOR CHRONIC HEPATITIS C
Sign Up to like & getrecommendations! Published in 2018 at "Journal of the American College of Cardiology"
DOI: 10.1016/s0735-1097(18)32304-0
Abstract: Some statins are not recommended for concomitant use with glecaprevir/pibrentasvir (G/P) treatment for chronic hepatitis C virus infection. We analyzed the impact of switching to rosuvastatin or pravastatin compared to maintaining or interrupting current statin… read more here.
Keywords: safety efficacy; chronic hepatitis; pibrentasvir treatment; glecaprevir pibrentasvir ... See more keywords

Early initiation of glecaprevir/pibrentasvir after transplantation of HCV‐viremic kidneys into HCV‐negative recipients is associated with normalization in the altered inflammatory milieu
Sign Up to like & getrecommendations! Published in 2023 at "Clinical Transplantation"
DOI: 10.1111/ctr.14926
Abstract: Our previous Multicenter Trial to Transplant HCV‐infected Kidneys (MYTHIC) observed that 100% of hepatitis C virus (HCV)‐uninfected patients who received a kidney from an HCV‐infected deceased donor were cured of HCV with an 8‐week regimen… read more here.
Keywords: glecaprevir pibrentasvir; inflammatory milieu; hcv viremic; hcv ... See more keywords
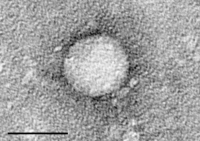
Retrospective multicentre study on the effectiveness of first-line direct-acting antivirals against hepatitis C virus genotype-1.
Sign Up to like & getrecommendations! Published in 2022 at "Journal of clinical pharmacy and therapeutics"
DOI: 10.1111/jcpt.13624
Abstract: WHAT IS KNOWN AND OBJECTIVE In Japan, ledipasvir/sofosbuvir, elbasvir/grazoprevir and glecaprevir/pibrentasvir are recommended as first-line treatments for patients with untreated hepatitis C virus genotype 1. Although they have demonstrated a high efficacy in clinical trials,… read more here.
Keywords: response; glecaprevir pibrentasvir; ledipasvir sofosbuvir; treatment ... See more keywords

Cell Culture Studies of the Efficacy and Barrier to Resistance of Sofosbuvir-Velpatasvir and Glecaprevir-Pibrentasvir against Hepatitis C Virus Genotypes 2a, 2b, and 2c
Sign Up to like & getrecommendations! Published in 2019 at "Antimicrobial Agents and Chemotherapy"
DOI: 10.1128/aac.01888-19
Abstract: The introduction of highly efficient therapies with direct-acting antivirals (DAA) for patients with chronic hepatitis C virus (HCV) infection offers exceptional opportunities to globally control this deadly disease. For achieving this ambitious goal, it is… read more here.
Keywords: glecaprevir pibrentasvir; resistance; virus; sofosbuvir ... See more keywords

Integrated Resistance Analysis of CERTAIN-1 and CERTAIN-2 Studies in Hepatitis C Virus-Infected Patients Receiving Glecaprevir and Pibrentasvir in Japan
Sign Up to like & getrecommendations! Published in 2017 at "Antimicrobial Agents and Chemotherapy"
DOI: 10.1128/aac.02217-17
Abstract: ABSTRACT Glecaprevir and pibrentasvir are hepatitis C virus (HCV) pangenotypic inhibitors targeting NS3/4A protease and NS5A, respectively. This once-daily, fixed-dose combination regimen demonstrated high sustained virologic response 12 weeks postdosing (SVR12) rates in CERTAIN-1 and… read more here.
Keywords: treatment; infected patients; certain certain; certain studies ... See more keywords

Glecaprevir/pibrentasvir+sofosbuvir: an optimal retreatment strategy in the setting of HCV NS5A resistance
Sign Up to like & getrecommendations! Published in 2020 at "BMJ Case Reports"
DOI: 10.1136/bcr-2019-233098
Abstract: Some individuals do not achieve a cure of their hepatitis C virus (HCV) infection due to non-adherence or resistance associated substitutions. Salvage options that are optimised for resistance profiles are essential. We report a 56-year-old… read more here.
Keywords: pibrentasvir sofosbuvir; hcv infection; sofosbuvir optimal; resistance ... See more keywords

Ledipasvir/Sofosbuvir is Effective for Relapsed Genotype 1b Hepatitis C Virus Patients after Achieving a Sustained Virological Response at Post-treatment Week 12 with Glecaprevir/Pibrentasvir.
Sign Up to like & getrecommendations! Published in 2022 at "Internal medicine"
DOI: 10.2169/internalmedicine.0865-22
Abstract: A patient with genotype 1b chronic hepatitis C virus who had been treated with pegylated interferon and ribavirin (RBV) was treated with glecaprevir/pibrentasvir (GLE/PIB) for 12 weeks. A sustained virological response at post-treatment week 12… read more here.
Keywords: sustained virological; glecaprevir pibrentasvir; hepatitis virus; treatment ... See more keywords

Risk Factors of Glecaprevir/Pibrentasvir-Induced Liver Injury and Efficacy of Ursodeoxycholic Acid
Sign Up to like & getrecommendations! Published in 2023 at "Viruses"
DOI: 10.3390/v15020489
Abstract: Although glecaprevir/pibrentasvir (GP) therapy is recommended as a first-line treatment for hepatitis C virus (HCV) infection, serious drug-induced liver injury occasionally develops. The present study aimed to elucidate real-world risk factors for GP-induced liver injury… read more here.
Keywords: glecaprevir pibrentasvir; liver injury; risk factors; ursodeoxycholic acid ... See more keywords

Efficacy and Safety of Glecaprevir/Pibrentasvir in Korean Patients with Chronic Hepatitis C: A Pooled Analysis of Five Phase II/III Trials.
Sign Up to like & getrecommendations! Published in 2021 at "Gut and liver"
DOI: 10.5009/gnl20321
Abstract: Background/Aims Glecaprevir/pibrentasvir (G/P) is the first pan-genotypic direct-acting antiviral combination therapy approved in Korea. An integrated analysis of five phase II and III trials was conducted to evaluate the efficacy and safety of G/P in… read more here.
Keywords: efficacy safety; analysis; treatment; safety ... See more keywords